The Real Opportunity of Precision Medicine and How to Not Miss Out
In his 2015 State of the Union address, President Obama announced a $215 million precision medicine initiative. At the time, many observers predicted that healthcare would heed his call and immediately get on board. Unlike the one-size-fits-all approach to medicine, precision medicine considers individual variability in genes, environment, and lifestyle to personalize care and greatly accelerate medical research and drug discoveries.
Improvements in health information technology have led to advances in genomics.
Emerging methods for managing and analyzing large data sets have empowered doctors and researchers to more accurately predict which treatment and prevention strategies for a particular disease will work in which groups of people.
What Is Precision Medicine?
Medical treatments are often designed for the so-called “average patient.” This one-size-fits-all approach to therapy can be successful for some patients, but not for others. President Obama’s Precision Medicine Initiative fact sheet defines precision medicine as “…a new model of patient-powered research that promises to accelerate biomedical discoveries and provide clinicians with new tools, knowledge, and therapies to select which treatments will work best for which patients.”
Put simply, precision medicine understands each patient’s individual illness and delivers the right treatment at the right time. Precision medicine integrates research with clinical practice. This cumulative knowledge empowers clinicians to create individualized care for each patient.
Clinical improvements to reduce system variation in care delivery can lead to high-quality health systems with lower cost for patients. In contrast, precision medicine seeks to understand pertinent patient variation from an individual’s unique environments and biochemistries. Reducing variation in healthcare is a good thing, but a provision for patient variation in treatments is also necessary.
Not all patients are the same. The challenge? Reconciling opposing views of variation in healthcare. The answer? Precise registries.
Precision Medicine Starts with Precise Registries
Remarkable advances in DNA sequencing technology are leading to the discovery of more diseases and enabling the development of treatments tailored to specific characteristics of individuals receiving the care. This molecular transformation of medicine is currently most visible in areas such as cancer, where patients with breast, lung, and colorectal cancers now undergo genetic testing as part of routine patient care. This permits the treating physicians to select a therapy ahead of time that will improve a patient’s chances of survival and reduce exposure to adverse effects. In essence, understanding the genetic variation removes the guesswork of choosing an effective therapy.
The key to leveraging this patient variation is precise registries. For example, instead of ICD-code based inclusion only, a more comprehensive registry would include keywords from problem lists, patients with medications specific to that condition, and other types of supplemental criteria. The difference in patient counts can often be pretty drastic as shown in Figure 1, where relying on the ICD-9 diagnosis code for asthma alone (n=29,805) compared to incorporating related terms and medications, yields a far more complete picture of patients and disease (n=101,389).

Precise definitions and registries of patients and disease are critical for clinical research, financial and risk management, personalized healthcare, and predictable outcomes. In the future, these definitions will become increasingly important in order for gene test results (and associated interpretations) to integrate with the EHR and drive actionable medical decisions.
Precise Medical Decision Making
We live in a remarkable era of information, when all that is known about a person—from family history and genetics to location history and environment—can be balanced against all that is known in the medical domain. This big-picture view of medical decision-making is allowing care providers to focus on prevention and intervention techniques on appropriate individuals, while avoiding unnecessary cost or unwanted side effects for those patients who would not benefit.
Traditional laboratory testing for point mutations and single gene assays are now being replaced by high-throughput gene panels, and exome or genome sequencing. Delivering gene test results, associated interpretation of gene variants, and appropriate clinical action in a timely manner becomes an important key to realizing the promise of precision medicine. (My colleagues and I published a study on this in the May 2012 edition of Genome Medicine called Consensus: a framework for evaluation of uncertain gene variants in laboratory test reporting.)
Academic Medical Centers Leading the Way
Mainstream EHR and health data warehouse offerings in the U.S. have begun to focus on capturing this genomic market segment. Recent advances in DNA sequencing technology have made the procedure, which cost up to $50,000 just a few years ago, relatively affordable at about $1,300, leading to broader use and important disease discoveries.
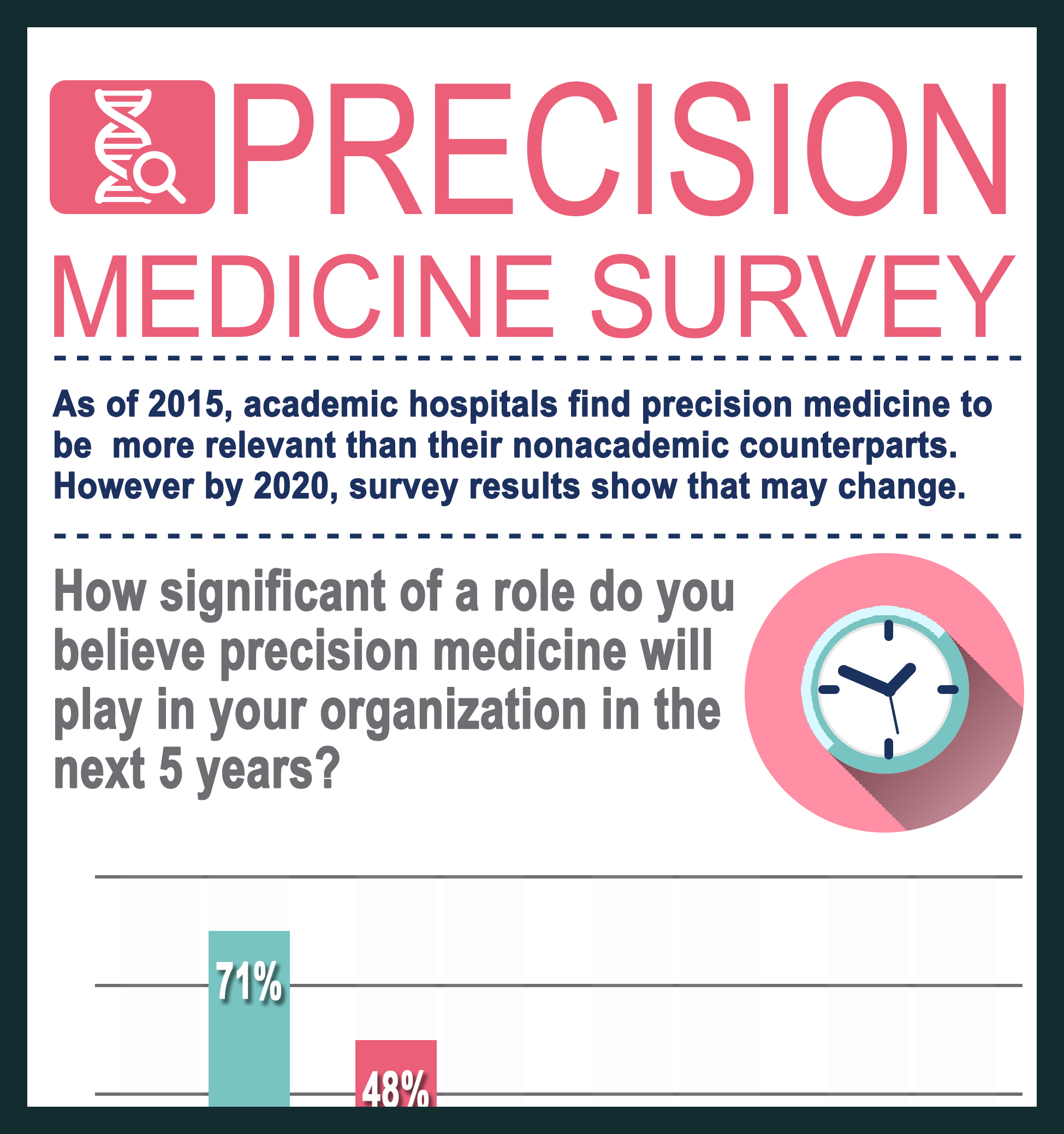
Despite this progress, few healthcare organizations are actively adopting precision medicine. The exception? Academic medical centers, which have historically led the adoption of new technologies in healthcare. In a recent survey, respondents from academic medical centers said precision medicine will play a significant role in their organizations in the next five years and they plan to integrate genomic data into their EHRs.
Experts liken this integration of genomics into the EHR to mapping terra incognita. The ability to practice precision medicine is largely dependent on gene variant knowledge bases, standard meta-data architecture, and decision-rule engines being made available to assist clinicians when taking action based on test results. In this important effort, the ability to link knowledge among databases ability to link knowledge among databases on genotype, phenotype, and clinical outcomes is crucial.
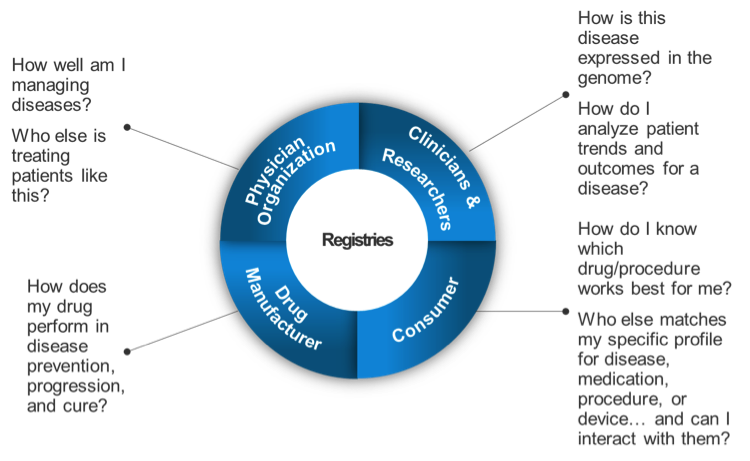
As shown in Figure 2, this integration of research data, clinical data, drug data, and patient data is the original motivation for building disease or patient registries. A precise registry can provide specific answers to specific audiences—even when the questions may not be anticipated ahead of time.
Precision medicine will continue to evolve. Precise patient registries will be required to ensure clinicians are armed with the most advanced tools, knowledge, and treatments that will work best for the patients in their care.
PowerPoint Slides
Would you like to use or share these concepts? Download this quality improvement presentation highlighting the key main points.
Click Here to Download the Slides
This website stores data such as cookies to enable essential site functionality, as well as marketing, personalization, and analytics. By remaining on this website you indicate your consent. For more information please visit our Privacy Policy.